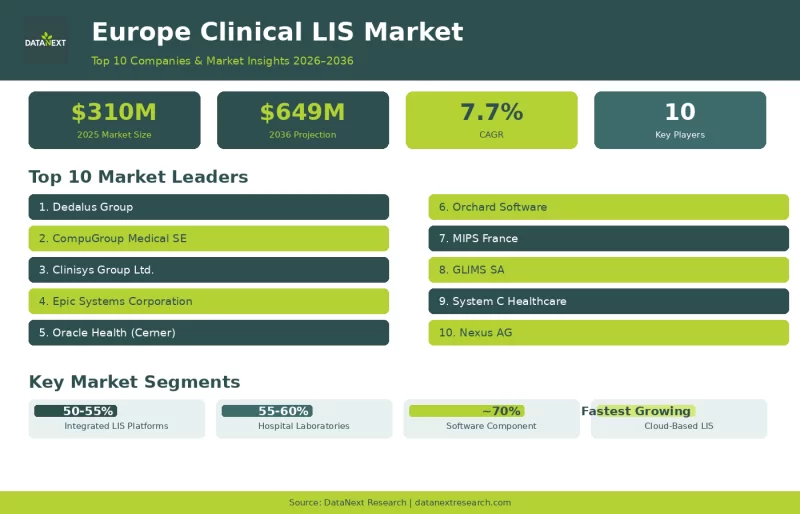
Top 10 Companies in the Europe Clinical Laboratory Information Systems (LIS) Market
At the heart of every hospital, diagnostic centre, and specialist laboratory in Europe lies an invisible infrastructure that makes modern medicine possible: the Laboratory Information System. Clinical LIS platforms are the digital nervous systems that track samples from the moment they arrive, manage testing workflows, validate results against quality benchmarks, interface with analyzers, and deliver reports to clinicians, all while maintaining the audit trails and data security standards demanded by European regulation. The Europe clinical LIS market was valued at USD 334 million in 2026 and is projected to reach USD 649 million by 2036, growing at a CAGR of 7.7%.
This growth is propelled by various factors. Diagnostic testing volumes are rising inexorably as populations age, chronic disease burdens increase, and personalized medicine drives demand for increasingly complex genomic and molecular assays. At the same time, the European Union's digital health agenda, anchored by the European Health Data Space regulation, national EHR mandates, and interoperability requirements under HL7 FHIR standards, is creating powerful regulatory pressure for laboratories to modernize legacy systems that can no longer meet the data-exchange, cybersecurity, and audit requirements of contemporary clinical practice.
Looking for a deeper understanding of how AI-enabled diagnostics, cloud-native LIS deployment, and healthcare data interoperability standards are shaping the future of laboratory digitalization across European healthcare systems?
Explore the full market analysis: https://datanextresearch.com/report/europe-clinical-laboratory-information-systems-lis-market
The shift from on-premise installations to cloud-native and hybrid deployment models is rewriting the economics of laboratory informatics, making enterprise-grade functionality accessible to independent and mid-sized laboratories that previously could not justify the capital outlay of traditional LIS implementations. Simultaneously, artificial intelligence is transforming the core functions of laboratory informatics, automating result validation, predicting instrument maintenance needs, flagging anomalous results for clinical review, and optimizing sample routing in high-throughput facilities. Here are the ten companies leading this transformation across European clinical laboratory sector.
1. Dedalus Group
Dedalus Group is one of Europe’s leading healthcare software providers and a major player in the region’s Clinical Laboratory Information Systems (LIS) market. Headquartered in Florence, Italy, the company has expanded its laboratory informatics capabilities through strategic acquisitions, including the healthcare software business of DXC Technology in 2020.
Dedalus offers laboratory information system solutions that are integrated with its broader Hospital Information System (HIS) and Electronic Patient Record (EPR) platforms. This integrated setup enables seamless data exchange between laboratories and clinical departments, helping healthcare providers manage patient test results, workflows, and reporting more efficiently.
The LIS platforms of the company are deployed across multiple European markets, including Italy, Germany, Austria, and the Benelux region, supporting hospital laboratories in areas such as clinical diagnostics, pathology, and molecular testing.
With operations in more than 40 countries and annual revenues approaching EUR 1 billion, Dedalus continues to invest in product localization, regulatory compliance, and customer support to meet the evolving digital healthcare infrastructure requirements of European healthcare providers.
2. CompuGroup Medical SE & Co. KGaA
CompuGroup Medical SE & Co. KGaA, headquartered in Koblenz, Germany, is one of Europe’s leading healthcare IT providers with a strong presence across the DACH region (Germany, Austria, and Switzerland), as well as expanding operations in Scandinavia, Benelux, and Southern Europe.
The company supports over 1.6 million healthcare professionals across more than 60 countries and generates annual revenues of approximately EUR 1.2 billion. Its laboratory informatics solutions are offered through the CGM LAB platform, which provides Laboratory Information System (LIS) capabilities for both hospital-based and independent diagnostic laboratories.
The LIS platform of CompuGroup Medical integrates with its broader healthcare IT ecosystem, including primary care and hospital information systems. This integration enables healthcare providers to manage test ordering, laboratory workflows, and result reporting through a unified digital interface.
The company has also expanded its cloud-based LIS offerings to support laboratory workflow optimization and improve turnaround times for clinical diagnostics. Ongoing investment in cloud infrastructure and digital tools positions CompuGroup Medical to support the increasing shift toward digitally connected laboratory environments across European healthcare systems.
3. Clinisys Group Ltd.
Clinisys Group Ltd. is a laboratory informatics company formed in 2022 through the merger of Sunquest Information Systems and CliniSys Solutions. The combined organization has strengthened its presence in the European Clinical Laboratory Information Systems (LIS) market by bringing together established laboratory software platforms and regional expertise.
One of the key LIS platforms of the company in Europe, WinPath Enterprise, is widely used across healthcare institutions in the United Kingdom, including NHS hospitals, independent pathology laboratories, and specialized diagnostic centers. The system supports multiple laboratory disciplines such as clinical chemistry, hematology, microbiology, immunology, and molecular diagnostics within a single digital workflow environment.
Clinisys also offers the GLIMS laboratory information platform, which has been adopted by healthcare providers in continental European markets including Belgium, the Netherlands, and France.
The company has expanded its LIS offerings to include cloud-based deployment options, allowing healthcare organizations to transition from on-premise systems to more scalable digital environments while maintaining existing laboratory workflows.
Through its laboratory informatics platforms, Clinisys supports diagnostic laboratories in managing test data, reporting, and operational workflows across hospital and independent laboratory networks in Europe.
4. Epic Systems Corporation
Epic Systems Corporation, headquartered in Verona, Wisconsin, is one of the world’s largest healthcare software providers and has established an expanding presence in the European Clinical Laboratory Information Systems (LIS) market through its Epic Beaker laboratory module.
While Epic is best known for its Electronic Health Records (EHR) platform used to manage clinical data for more than 300 million patient records globally, its Beaker LIS solution has gained adoption among European hospital networks undertaking enterprise-wide digital transformation initiatives.
The primary competitive strength of Epic Beaker lies in its native integration within Epic’s broader clinical platform, enabling laboratory test orders to be generated directly from physician workflows and validated results to be automatically updated in the patient’s electronic health record. This integrated architecture reduces the need for third-party middleware typically required when deploying standalone LIS platforms alongside separate EHR systems.
The European footprint of Epic has expanded across major healthcare markets including the United Kingdom, the Netherlands, Denmark, and Finland, where academic medical centres and integrated hospital systems are implementing unified clinical information platforms for long-term digital infrastructure planning.
Through its enterprise-wide approach to healthcare IT deployment, Epic supports laboratory operations within a consolidated digital environment that combines patient records, diagnostic workflows, and clinical communication systems.
5. Oracle Health (formerly Cerner Corporation)
Oracle Health, established following Oracle Corporation’s acquisition of Cerner Corporation in a USD 28.3 billion transaction completed in 2022, combines Cerner’s clinical informatics expertise with Oracle’s cloud infrastructure and enterprise software capabilities.
Within the European Clinical Laboratory Information Systems (LIS) market, Oracle Health offers laboratory informatics solutions through Cerner’s established platforms, including integrated LIS functionality within the PowerChart Electronic Health Record (EHR) environment and the standalone PathNet laboratory information system for healthcare providers not operating the full Cerner clinical suite.
The PathNet platform supports a wide range of laboratory disciplines, with particular strength in anatomic pathology and molecular diagnostics workflows, including case management, result interpretation, and digital image integration. The system is also compatible with digital pathology technologies such as whole-slide imaging platforms from manufacturers including Leica, Hamamatsu, and Philips.
Oracle Health continues to expand cloud-based deployment pathways for existing Cerner LIS customers, enabling healthcare organizations to transition from traditional on-premise systems to more scalable digital environments.
Backed by global enterprise software ecosystem of Oracle and ongoing investment in healthcare IT infrastructure, Oracle Health remains a key participant in the European laboratory informatics market supporting hospital and independent diagnostic laboratory operations.
6. Orchard Software (Orchard Systems)
Orchard Software, headquartered in Indiana, United States, is a laboratory informatics provider with an expanding footprint in the European Clinical Laboratory Information Systems (LIS) market. The company’s Orchard Harvest™ LIS platform is widely used by independent diagnostic laboratories, specialty testing facilities, and physician office laboratories requiring configurable workflow management solutions.
A key strength of Orchard Harvest lies in its instrument interfacing capabilities, supporting bidirectional communication with more than 700 laboratory analyzer models from major manufacturers including Roche, Abbott, Siemens, Beckman Coulter, and Bio-Rad. This enables automated test result transfer, calibration monitoring, and quality control management—helping reduce manual data entry and improve operational efficiency within laboratory workflows.
The platform also incorporates configurable rules-based validation tools that allow routine test results to be automatically reviewed based on predefined criteria, while flagging atypical or critical values for manual assessment.
Orchard Software currently supports over 800 laboratory clients globally, with its European expansion strategy focused on independent diagnostic groups and specialty laboratories seeking scalable LIS solutions without the complexity associated with enterprise-level hospital information systems.
Through its configurable system architecture and established deployment base, Orchard Software continues to support laboratory digitalization initiatives across mid-sized and independent healthcare providers in Europe.
7. MIPS France
MIPS France, part of the international MIPS healthcare software group headquartered in Sweden, maintains an established presence in the French Clinical Laboratory Information Systems (LIS) market through its Kalisil laboratory information platform.
The Kalisil LIS is designed to support the regulatory and operational requirements of clinical laboratories operating within France’s national healthcare framework. The system includes functionality aligned with ISO 15189 accreditation standards for medical laboratories, enabling laboratories to manage quality control processes, external quality assessments, non-conformity tracking, and audit documentation required for accreditation by the Comité français d'accréditation (COFRAC).
The platform is also integrated to support interoperability with France’s national healthcare data infrastructure, including connectivity to the Mon espace santé patient health record system.
MIPS France has expanded its compliance capabilities in line with the Ségur du numérique healthcare digitalization initiative, incorporating interoperability standards such as HL7 FHIR to support secure health data exchange across connected healthcare IT platforms.
Through its regulatory-aligned LIS solutions, MIPS France supports accredited clinical laboratories in managing diagnostic workflows and quality documentation within France’s evolving digital healthcare ecosystem.
8. GLIMS SA (Now part of Clinisys)
GLIMS SA, originally developed in Belgium and now operating as part of the Clinisys Group Ltd. following its 2022 integration, remains a widely deployed laboratory information system across European healthcare networks.
The GLIMS (Global Laboratory Information Management System) platform is designed to support multi-site laboratory operations, enabling healthcare providers to manage diagnostic workflows, sample tracking, and result reporting across geographically distributed laboratory facilities through a unified digital environment.
This architecture is particularly relevant for hospital networks operating in hub-and-spoke diagnostic models, where centralized reference laboratories perform specialized testing while satellite laboratories manage routine diagnostic workloads.
GLIMS includes workflow management tools for monitoring sample transport logistics, turnaround times, and quality control processes across laboratory networks, supporting operational consistency across multiple sites.
The platform has been adopted by hospital networks across Belgium, the Netherlands, and France, including installations in university hospital environments requiring integrated laboratory data management across clinical disciplines.
Under Clinisys ownership, GLIMS continues to benefit from investment in cloud deployment capabilities and interoperability with other laboratory informatics platforms within the Clinisys portfolio, supporting digital transformation initiatives across European diagnostic services.
9. System C Healthcare
System C Healthcare, headquartered in the United Kingdom, is a healthcare IT provider with more than four decades of experience supporting digital infrastructure within the National Health Service (NHS). The company currently serves over 100 NHS organizations across the UK through its integrated clinical information platforms.
The laboratory informatics capabilities of System C are delivered as part of its Maxims digital health system, which combines electronic patient records (EPR), clinical decision support tools, and laboratory management functionality within a unified data architecture.
This integrated platform enables seamless communication between diagnostic laboratories and clinical departments, reducing reliance on third-party interoperability solutions typically required when deploying standalone Laboratory Information Systems (LIS).
System C has also expanded its cloud-based deployment capabilities in line with NHS England’s Cloud First policy, supporting healthcare providers in transitioning toward cloud-hosted digital health environments for clinical and diagnostic system management.
Through its long-standing engagement with NHS digital transformation programmes, System C supports laboratory workflow management, patient data integration, and compliance with clinical governance requirements across hospital and independent healthcare settings in the UK.
10. Nexus AG
Nexus AG, headquartered in Villingen-Schwenningen, Germany, is a healthcare IT company with a strong presence across the DACH region (Germany, Austria, and Switzerland). The company offers laboratory informatics solutions through its NEXUS / LABOR Laboratory Information System (LIS), designed to support the quality and documentation requirements of accredited clinical laboratories.
NEXUS / LABOR is integrated with the company’s broader healthcare IT portfolio, including the NEXUS / KIS hospital information system and the NEXUS / PACS imaging platform. This integration enables laboratory test ordering directly from clinical departments, automated result reporting to electronic patient records, and centralized audit documentation for accreditation purposes.
Nexus AG currently serves over 1,000 healthcare facilities and generates annual revenues of approximately EUR 200 million. The company has expanded its cloud deployment capabilities in alignment with Germany’s Telematikinfrastruktur national healthcare data network. Additionally, NEXUS / LABOR supports digital healthcare modernization initiatives under Germany’s Hospital Future Act (Krankenhauszukunftsgesetz), which encourages hospitals and diagnostic laboratories to adopt hybrid and cloud-enabled IT systems.
Through its integrated platform approach and localized regulatory alignment, Nexus AG continues to support laboratory digitalization across healthcare providers in German-speaking European markets.
The Digital Backbone of European Healthcare Diagnostics
The Europe Clinical Laboratory Information Systems (LIS) market is projected to grow from USD 334 million in 2026 to USD 649 million by 2036, reflecting more than an increase in software adoption. This growth signals a broader shift in how healthcare providers across Europe manage and utilize diagnostic data within clinical decision-making processes.
Laboratory test results, which were once managed as standalone reports, are increasingly being integrated into connected patient data systems. These systems allow diagnostic information to be securely accessed, shared, and analyzed across departments, supporting coordinated patient care across hospitals, outpatient clinics, and specialist laboratories.
The ten companies profiled in this report illustrate the structure of this evolving market. European healthcare IT providers such as Dedalus Group and CompuGroup Medical bring localized regulatory expertise and established hospital network relationships. Specialist laboratory informatics providers such as Clinisys and GLIMS support multi-site laboratory networks and centralized diagnostic workflows. Global enterprise platform vendors including Epic Systems and Oracle Health offer LIS capabilities integrated within broader electronic health record environments. Orchard Software addresses the needs of independent laboratories through configurable instrument integration platforms, while regionally focused providers such as MIPS France and Nexus AG support domestic compliance and accreditation requirements. System C Healthcare contributes operational expertise aligned with the UK’s NHS digital infrastructure.
Looking ahead, the adoption of cloud-based LIS deployment models, AI-supported diagnostic workflows, and HL7 FHIR interoperability standards is expected to become increasingly common across European healthcare systems. Vendors that combine scalable digital platforms with regulatory compliance, localized implementation support, and long-term service capabilities will be well positioned to support laboratory digitalization initiatives through the forecast period.
With hospital laboratories across Europe increasingly investing in advanced LIS platforms to improve diagnostic accuracy, workflow efficiency, and regulatory compliance, digital laboratory informatics is becoming central to modern clinical decision-making.
Are you positioned to benefit from this transition?
Our Europe Clinical Laboratory Information Systems (LIS) Market (2026–2036) report delivers:
- Country-level LIS adoption outlook
- Cloud vs. on-premise deployment trends
- AI-assisted diagnostic workflow integration
- HL7 FHIR interoperability landscape
- Competitive profiling of leading LIS vendors
Download the Sample Report Now:
https://datanextresearch.com/report/europe-clinical-laboratory-information-systems-lis-market?action=Download+Sample
Access the Full Market Report:
https://datanextresearch.com/report/europe-clinical-laboratory-information-systems-lis-market
Related Articles
Top 10 Companies in the Southeast Asia Smart Syringe Market
Discover the Top 10 Companies in the Southeast Asia Smart Syringe Market driving growth to USD 1.47B by 2036, fueled by safety innovation, immunization demand, and healthcare modernization.

Top 10 Biotech Startups in Europe to Watch (2026)
Discover the top 10 biotech startups in Europe driving innovation in 2026. Explore breakthroughs in cancer therapies, AI-driven drug discovery, genomics, and cell reprogramming, backed by strong funding and EU support.
