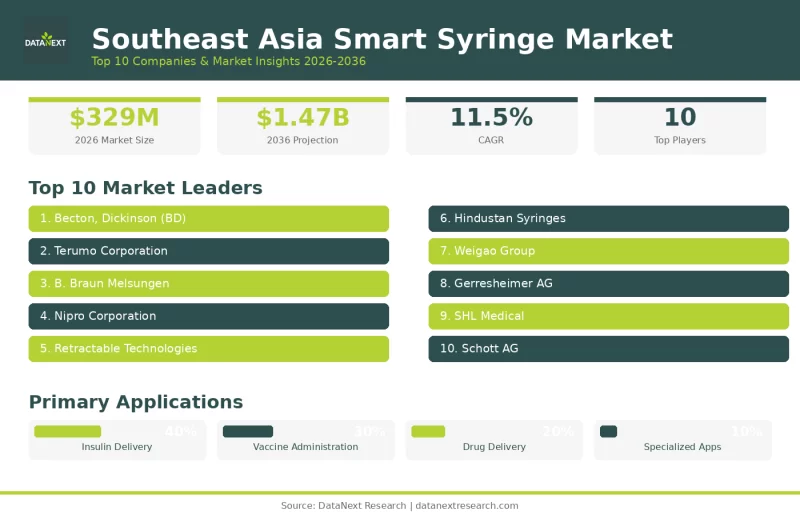
Top 10 Companies in the Southeast Asia Smart Syringe Market
The Southeast Asia smart syringe market is experiencing significant growth, driven by increasing healthcare modernization, rising chronic disease prevalence, and heightened focus on medication safety. Smart syringes, which incorporate features like safety mechanisms, precise dose delivery, tracking capabilities, and reuse prevention, represent a critical evolution in medical device technology. These advanced delivery systems are becoming essential tools in the healthcare infrastructure, supporting everything from mass immunization programs to chronic disease management in this region.
The expansion of this market reflects broader healthcare priorities across Southeast Asia. With diabetes rates climbing, extensive vaccination campaigns underway, and healthcare systems modernizing rapidly, smart syringes offer solutions that improve both patient safety and treatment outcomes. According to the market report published by DataNext Research, the Southeast Asia smart syringe market is projected to grow from USD 329 million in 2026 to USD 1.47 billion by 2036, at a CAGR of 11.5%. Thailand currently leads the regional market, while Vietnam shows the fastest growth, supported by rising healthcare investments and increasing disease prevalence.
Looking for a deeper understanding of how infection-prevention policies, mass immunization programs, and safety-engineered injection technologies are shaping the future of smart syringe adoption across Southeast Asia?
Explore the full market analysis: https://datanextresearch.com/report/southeast-asia-smart-syringe-market
This growth is being shaped by innovative companies that are advancing smart syringe technology, expanding manufacturing capacity, and developing integrated healthcare solutions. Here are the top 10 companies driving this transformation in Southeast Asia smart syringe market.
1. Becton, Dickinson and Company (BD)
Becton, Dickinson and Company (BD) is one of the leading manufacturers of safety-engineered injection devices and smart syringe systems used in immunization and clinical drug administration programs globally. The company offers a wide range of injection technologies through its BD Medical–Pharmaceutical Systems segment, including auto-disable (AD) syringes and safety syringes designed to prevent reuse and reduce the risk of needlestick injuries in healthcare settings.
The smart syringe portfolio of BD is widely utilized in vaccination programs supported by international health organizations across Southeast Asia, particularly for routine immunization and public health initiatives aimed at reducing the transmission of bloodborne infections. The deployment of auto-disable syringe technologies has been instrumental in supporting safe injection practices in countries such as Indonesia, Vietnam, and the Philippines, where large-scale immunization campaigns are conducted through government-led healthcare programs.
The company markets its injection devices in over 190 countries worldwide and maintains a strong distribution presence across Asia-Pacific through regional supply networks. BD’s product portfolio supporting insulin delivery, vaccine administration, and clinical drug delivery positions the company as a key supplier of safety-engineered injection systems across Southeast Asia’s public and private healthcare infrastructure.
2. Terumo Corporation
Terumo Corporation, founded in 1921 in Japan, is a global manufacturer of injection and infusion therapy devices with an established presence across Southeast Asia. The company initially focused on clinical thermometer production and expanded its product portfolio to include disposable syringes, becoming one of the early manufacturers to introduce disposable syringe systems in Japan in 1963.
Terumo offers a range of safety-engineered injection devices, insulin delivery systems, and prefillable syringe platforms for pharmaceutical and clinical applications. The company’s PLAJEX® ready-to-fill polymer syringe system is designed for biologics and specialty drug delivery and is manufactured using cyclo-olefin polymer (COP), providing silicone oil-free primary packaging for injectable therapies.
The injection devices of Terumo are widely utilized in hospital and immunization settings across Southeast Asia for vaccine administration and diabetes management. The company maintains a regional distribution network across Asia-Pacific and operates medical device manufacturing facilities compliant with international quality management standards such as ISO 13485. Terumo’s product portfolio and regional supply capabilities position the company as a key supplier of safety-engineered injection systems across Southeast Asia’s healthcare infrastructure.
3. B. Braun Melsungen AG
B. Braun Melsungen AG is a Germany-based medical device manufacturer offering injection and infusion therapy systems for clinical and immunization applications. Founded in 1839, the company operates in more than 60 countries worldwide and employs over 60,000 people across its global operations. B. Braun’s product portfolio includes safety-engineered syringes, insulin delivery devices, infusion systems, and medication delivery platforms used across hospital and public healthcare settings.
The syringe portfolio of the company includes the Omnifix® and Injekt® product lines, which are utilized for drug administration and vaccine delivery across healthcare facilities. B. Braun also offers Original Perfusor® syringes designed for compatibility with automated syringe pumps used in controlled infusion therapy.
B. Braun maintains a presence across Asia-Pacific through regional manufacturing and distribution networks supporting the supply of injection devices to hospital systems and immunization programs. The company operates medical device manufacturing facilities compliant with international quality management standards such as ISO 13485. B. Braun’s injection and infusion therapy solutions are utilized across Southeast Asia for routine clinical drug administration and vaccination programs, supporting safe injection practices across public and private healthcare settings.
4. Nipro Corporation
Nipro Corporation is a Japan-based medical device manufacturer offering a range of injection and infusion therapy products for clinical and pharmaceutical applications. The company provides disposable syringes, insulin delivery devices, safety syringes, and prefillable syringe systems used for drug administration and immunization programs.
Nipro’s smart syringe portfolio includes auto-disable syringes designed to prevent reuse and support safe injection practices in healthcare settings. The company also offers
Direct-to-Fill (D2F®) prefillable glass syringe systems for pharmaceutical applications, supporting the delivery of biologics and specialty injectable therapies.
Nipro operates manufacturing and distribution facilities across Asia, Europe, and North America, enabling regional supply capabilities for healthcare providers and immunization programs. The company’s injection devices are utilized across hospital systems and public health initiatives in Southeast Asia for routine clinical drug administration and vaccination programs. In addition to syringe systems, Nipro’s broader product portfolio includes IV catheters, infusion sets, and blood collection systems, supporting integrated medication delivery solutions across healthcare settings in the region.
5. Retractable Technologies, Inc.
Retractable Technologies, Inc. is a U.S.-based manufacturer of safety-engineered injection devices specializing in retractable syringe technologies designed to reduce needlestick injuries and prevent syringe reuse. Founded in 1994 and headquartered in Little Elm, Texas, the company develops safety syringes, insulin syringes, blood collection devices, and IV catheter products for clinical and immunization applications.
The VanishPoint® safety syringe platform of the company features automated needle retraction mechanisms that activate upon completion of the injection process, thereby minimizing the risk of needlestick injuries and enhancing injection safety in healthcare settings. Retractable Technologies also offers low dead space syringe configurations used in vaccination programs to support dose efficiency during immunization campaigns.
Retractable Technologies manufactures safety-engineered injection devices for distribution across global healthcare markets. In Southeast Asia, the company’s retractable syringe technologies are utilized in hospital-based drug administration and vaccination programs aimed at improving safe injection practices and reducing the risk of bloodborne infections. The company’s product portfolio positions it as a supplier of smart syringe solutions supporting public health initiatives across the region.
6. Hindustan Syringes & Medical Devices Ltd. (HMD)
Hindustan Syringes & Medical Devices Ltd. (HMD), established in 1957 and headquartered in Faridabad, India, is a manufacturer of disposable syringes and safety-engineered injection devices supplying domestic and international healthcare markets, including Southeast Asia. The company produces a wide range of single-use syringes under brands such as Dispovan® and Unolok®, along with auto-disable (AD) syringes designed for immunization programs and insulin delivery systems for diabetes management.
HMD manufactures billions of injection devices annually across its integrated production facilities and supplies safety syringes used in vaccination and routine drug administration across public healthcare systems. The company expanded its production capacity during the COVID-19 pandemic to support immunization campaigns, including the supply of low dead space and auto-disable syringes for vaccine delivery.
HMD operates manufacturing facilities compliant with international quality management standards such as ISO 13485, enabling the supply of injection devices to healthcare providers across global markets. In Southeast Asia, the company’s safety-engineered syringes are utilized in government-led immunization initiatives and hospital-based drug administration programs, supporting safe injection practices across the region’s healthcare infrastructure.
7. Weigao Group Medical Polymer Company Limited
Shandong Weigao Group Medical Polymer Company Limited is a China-based manufacturer of medical device consumables, including disposable syringes and safety-engineered injection systems. Headquartered in Weihai and listed on the Hong Kong Stock Exchange since 2004, the company supplies a wide range of medical products used in hospital-based drug administration and immunization programs.
The injection device portfolio of the company includes disposable syringes, auto-disable (AD) syringes, medical needles, and prefillable syringe systems used for vaccine delivery and pharmaceutical applications. These products are utilized in clinical and public healthcare settings to support safe injection practices and reduce the risk of syringe reuse.
The company operates manufacturing facilities compliant with international quality management standards such as ISO 13485, enabling the supply of medical consumables across global healthcare markets. While China remains its primary market, Weigao is expanding its presence in Asia-Pacific through regional distribution networks. In Southeast Asia, the company’s safety syringe products are deployed across immunization initiatives and hospital-based drug administration programs, positioning it as a supplier of smart syringe solutions across the region’s healthcare infrastructure.
8. Gerresheimer AG
Gerresheimer AG is a Germany-based manufacturer of pharmaceutical primary packaging and drug delivery systems, supplying prefillable syringe platforms for injectable therapies. The company operates more than 40 production facilities globally and employs over 11,000 people across Europe, North America, Asia, and South America.
The syringe portfolio of Gerresheimer includes the Gx RTF® (Ready-to-Fill) platform, comprising Gx Value RTF®, Gx Performance RTF®, and Gx Elite RTF® syringes designed for pharmaceutical and biopharmaceutical applications. The company also offers ClearJect® polymer syringes manufactured using cyclo-olefin polymer (COP), supporting the delivery of sensitive biologic drugs and vaccines.
Gerresheimer develops silicone oil–reduced and silicone oil–free prefillable syringe systems to support drug stability and minimize particulate contamination during injectable drug administration. These systems are utilized by pharmaceutical companies for the delivery of vaccines and specialty therapeutics through prefilled injection formats.
Gerresheimer maintains manufacturing and development capabilities across Asia, supporting pharmaceutical packaging supply chains across emerging healthcare markets. In Southeast Asia, the company’s prefillable syringe platforms are utilized for vaccine and biologic drug delivery, positioning it as a supplier of smart syringe systems for pharmaceutical applications across the region.
9. SHL Medical
SHL Medical is a Switzerland-based developer and manufacturer of drug delivery devices, specializing in autoinjectors, pen injectors, and wearable injection systems for pharmaceutical applications. Founded in 1989, the company provides injection platforms designed for the subcutaneous administration of biologics and specialty therapeutics.
The product portfolio of SHL Medical includes autoinjector platforms such as Molly® and Maggie®, which are designed for compatibility with prefilled syringe systems used in the delivery of chronic disease therapies. These devices support self-administration of injectable drugs across therapeutic areas such as diabetes, autoimmune disorders, and cardiovascular diseases.
The company collaborates with pharmaceutical manufacturers to provide device development and assembly services for combination drug–device products. SHL Medical operates manufacturing and development facilities across Europe, Asia, and North America, supporting global pharmaceutical supply chains.
In Southeast Asia, SHL Medical’s autoinjector platforms are utilized for the delivery of biologic therapies through prefilled injection formats, supporting the growing adoption of advanced drug delivery systems for chronic disease management across the region’s healthcare infrastructure.
10. Schott AG
SCHOTT AG, a subsidiary of the Carl Zeiss Stiftung, is a Germany-based manufacturer of specialty glass used in pharmaceutical primary packaging applications. Through its SCHOTT Pharma business, the company supplies prefillable glass syringe systems designed for injectable drug delivery, along with vials, ampoules, and cartridges used in pharmaceutical manufacturing.
The syrinX® prefillable syringe platform of SCHOTT is manufactured using Type I borosilicate glass and is utilized in the delivery of vaccines, biologics, and specialty injectable therapies. These syringe systems are designed to support automated filling processes and maintain container closure integrity for pharmaceutical applications.
SCHOTT operates production and development facilities across Europe, North America, and Asia, supporting global pharmaceutical supply chains. In Southeast Asia, the company provides technical and commercial support to pharmaceutical manufacturers utilizing prefillable syringe platforms for vaccine and biologic drug delivery. SCHOTT’s primary packaging solutions enable the deployment of prefilled injection systems across the region’s healthcare infrastructure.
Shaping the Future of Healthcare Delivery
The Southeast Asia smart syringe market is mainly driven by various factors such as rising chronic disease prevalence, healthcare modernization initiatives, increased focus on medication safety, and growing adoption of self-administration therapies. These top 10 companies are not merely responding to market opportunities but actively shaping the future of how medications are delivered throughout the region.
Each company brings distinct strengths to the competitive landscape. Global giants like BD and Terumo leverage extensive manufacturing scale, broad product portfolios, and deep market penetration. German precision manufacturers like B. Braun, Gerresheimer, and Schott contribute engineering excellence and quality standards. Japanese companies Terumo and Nipro offer technological innovation combined with Asian market understanding. American innovator Retractable Technologies advances safety technology. Indian manufacturer HMD provides cost-effective solutions at scale. Chinese manufacturer Weigao represents growing domestic capability. Swiss specialist SHL Medical pushes the boundaries of connected drug delivery systems.
The projected growth of this market to USD 1.47 billion by 2036 will be driven by these companies' continued investments in innovation, manufacturing capacity, and market development. Smart syringes incorporating artificial intelligence, advanced connectivity, medication verification, and comprehensive safety management represent the next frontier. As Southeast Asian healthcare systems continue modernizing and patients increasingly manage chronic conditions at home, these companies will play essential roles in ensuring safe, effective, and accessible medication delivery across the region.
The competitive dynamics will likely intensify as companies balance innovation with affordability, global capabilities with local market knowledge, and advanced features with user simplicity. Success in Southeast Asia's diverse markets will require not just superior products but also comprehensive support services, regulatory expertise, and deep understanding of local healthcare delivery challenges. The companies that can deliver on these multiple dimensions will be best positioned to capture value in this rapidly expanding market.
Looking for a deeper understanding of how safety regulations, vaccination programs, and rising demand for infection-prevention technologies are shaping the future of smart syringe adoption across Southeast Asia?
Explore the full market analysis: https://datanextresearch.com/report/southeast-asia-smart-syringe-market
You can also download a complimentary sample here:
https://datanextresearch.com/report/southeast-asia-smart-syringe-market?action=Download+Sample
Related Articles
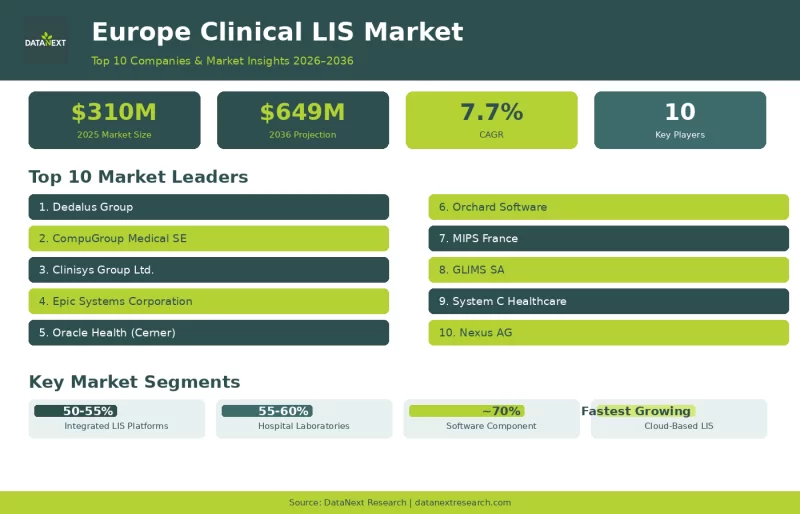
Top 10 Companies in the Europe Clinical Laboratory Information Systems (LIS) Market
Discover the Top 10 Companies in the Europe Clinical Laboratory Information Systems (LIS) Market driving growth with AI diagnostics, cloud LIS, and interoperable healthcare platforms.

Top 10 Biotech Startups in Europe to Watch (2026)
Discover the top 10 biotech startups in Europe driving innovation in 2026. Explore breakthroughs in cancer therapies, AI-driven drug discovery, genomics, and cell reprogramming, backed by strong funding and EU support.
